狄诺塞麦注射剂Xgeva Injektionslösung 120mg/1.7ml
[caption id="attachment_19873" align="alignleft" width="300"]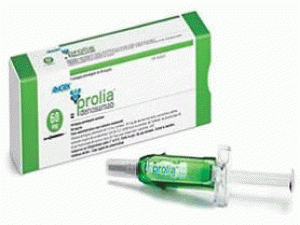 狄诺塞麦注射剂Xgeva Injektionslösung 120mg/1.7ml[/caption]
药店国别:
产地国家: 瑞士
处方药:是
所属类别: 120毫克/1.7毫升/瓶
包装规格:120毫克/1.7毫升/瓶
计价单位:瓶
生产厂家中文参考译名:安进瑞士AG
生产厂家英文名:Amgen Switzerland AG
原产地英文商品名:Xgeva Inj Loes 120mg/1.7mL/Vial
原产地英文药品名:denosumab
中文参考商品译名:Xgeva注射剂 120毫克/1.7毫升/瓶
中文参考药品译名:Xgeva注射剂 120毫克/1.7毫升/瓶
曾用名:
简介:
部份中文狄迪诺赛麦处方资料(仅供参考)英文药名:Xgeva英文名称:denosumab中文药名:狄迪诺赛麦注射剂药品介绍:Xgeva(狄迪诺塞麦)是一种RANK配体的抑制剂,由Amgen公司生产,2010年11月18日批准用于防止已扩散到骨的癌症,2013年6月13日批准用于治疗骨巨细胞瘤(GCTB)成年和青少年,一种罕见和通常非癌性肿瘤。
适应症:
对有实体瘤且转移的患者预防,狄迪诺赛麦不适合用于多发性骨髓瘤的患者为预防骨骼做预防。
用法用量
Xgeva仅供皮下途径而不应被静脉内,肌内,或皮内骨转移从实体瘤:管理120毫克每4周作为皮下注射在上臂,大腿上部,或腹部骨巨细胞瘤:辖120毫克,每4周增加120毫克剂量的8天15在第一个月的治疗。皮下注射,在上臂,大腿上部,或腹部管理钙和维生素D是必要的治疗或预防低钙血症恶性高钙血症:辖120毫克,每4周增加120毫克剂量的8天15在第一个月的治疗。皮下注射,在上臂,大腿上部,或腹部
狄诺塞麦注射剂英文版说明书
Safety InformationDo not take XGEVA® if you have low blood calcium (hypocalcemia). Your low blood calcium must be treated before you receive XGEVA®. XGEVA® can significantly lower the calcium levels in your blood and some deaths have been reported. Take calcium and vitamin D as your doctor tells you to. Tell your doctor right away if you experience spasms, twitches, cramps, or stiffness in your muscles or numbness or tingling in your fingers, toes, or around your mouth.Do not take XGEVA® if you are allergic to denosumab or any of the ingredients of XGEVA®. Serious allergic reactions have happened in people who take XGEVA®. Call your doctor or go to your nearest emergency room right away if you have any symptoms of a serious allergic reaction, including low blood pressure (hypotension); trouble breathing; throat tightness; swelling of the face, lips, or tongue, rash; itching; or hives.What is the most important information you should know about XGEVA®?Do not take XGEVA® if you take Prolia. XGEVA® contains the same medicine as Prolia® (denosumab).Severe jaw bone problems (osteonecrosis)Severe jaw bone problems may happen when you take XGEVA®. Your doctor should examine your mouth before you start, and while you are taking XGEVA®. Tell your dentist that you are taking XGEVA®. It is important for you to practice good mouth care during treatment with XGEVA®. In studies of patients with bone involvement, the rate of severe jaw problems was higher the longer they were being treated with XGEVA®.Unusual thigh bone fractureUnusual thigh bone fracture has been reported. Symptoms of a fracture include new or unusual pain in your hip, groin, or thigh.Risk of high calcium levels in patients with Giant Cell Tumor of Bone and in patients who are still growingPatients with a type of cancer called Giant Cell Tumor of Bone and patients with bones that are not fully matured are at a greater risk to develop high blood calcium levels after they stop taking XGEVA®, that can be serious.Increased risk of broken bones in the spine after discontinuing XGEVA®After your treatment with XGEVA® is stopped, your risk for breaking bones in your spine can increase, especially if you have a history of risk factors such as osteoporosis or prior fractures.Possible harm to your unborn babyYou should not become pregnant while taking XGEVA®. Tell your doctor right away if you are pregnant, plan to become pregnant, or suspect you are pregnant. XGEVA® can harm your unborn baby.Tell your doctor if you:Are taking a medicine called Prolia® (denosumab) because it contains the same medicine as XGEVA®Have symptoms of low blood calcium such as muscle stiffness or crampsHave symptoms of severe jaw bone problems such as pain or numbnessHave ongoing pain or slow healing after dental surgeryHave symptoms of high blood calcium such as nausea, vomiting, headache, and decreased alertnessAre pregnant, plan to become pregnant, suspect you are pregnant, or breastfeedingWhile taking XGEVA®, you should:Take good care of your teeth and gums and visit a dentist as recommendedTell your dentist that you are taking XGEVA®Tell your doctor if you plan to have dental surgery or teeth removedTalk to your doctor before you stop taking XGEVA® about your risk for broken bones in your spineWomen of child bearing age should use highly effective contraception while taking XGEVA® and for at least 5 months after the last dose of XGEVA®What are the possible side effects of XGEVA®?In patients with bone metastases from solid tumors using XGEVA®, the most common side effects were tiredness/weakness, low phosphate levels in your blood, and nausea. The most common serious side effect of XGEVA® was shortness of breath.In multiple myeloma patients receiving XGEVA®, the most common side effects were diarrhea, nausea, low red blood cells, low blood platelets and calcium levels, back pain, swelling of the lower legs or hands, upper respiratory tract infection, rash, and headache. The most common serious adverse reaction in multiple myeloma patients was pneumonia.These are not all the possible side effects of XGEVA®. For more information, ask your doctor or pharmacist.Call your doctor for medical advice about side effects. You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.Please see Full Prescribing Information.IndicationXGEVA® is a prescription medicine used to prevent fracture, spinal cord compression, or the need for radiation or surgery to bone in patients with multiple myeloma and in patients with bone metastases from solid tumors.
狄诺塞麦注射剂Xgeva Injektionslösung 120mg/1.7ml[/caption]
药店国别:
产地国家: 瑞士
处方药:是
所属类别: 120毫克/1.7毫升/瓶
包装规格:120毫克/1.7毫升/瓶
计价单位:瓶
生产厂家中文参考译名:安进瑞士AG
生产厂家英文名:Amgen Switzerland AG
原产地英文商品名:Xgeva Inj Loes 120mg/1.7mL/Vial
原产地英文药品名:denosumab
中文参考商品译名:Xgeva注射剂 120毫克/1.7毫升/瓶
中文参考药品译名:Xgeva注射剂 120毫克/1.7毫升/瓶
曾用名:
简介:
部份中文狄迪诺赛麦处方资料(仅供参考)英文药名:Xgeva英文名称:denosumab中文药名:狄迪诺赛麦注射剂药品介绍:Xgeva(狄迪诺塞麦)是一种RANK配体的抑制剂,由Amgen公司生产,2010年11月18日批准用于防止已扩散到骨的癌症,2013年6月13日批准用于治疗骨巨细胞瘤(GCTB)成年和青少年,一种罕见和通常非癌性肿瘤。
适应症:
对有实体瘤且转移的患者预防,狄迪诺赛麦不适合用于多发性骨髓瘤的患者为预防骨骼做预防。
用法用量
Xgeva仅供皮下途径而不应被静脉内,肌内,或皮内骨转移从实体瘤:管理120毫克每4周作为皮下注射在上臂,大腿上部,或腹部骨巨细胞瘤:辖120毫克,每4周增加120毫克剂量的8天15在第一个月的治疗。皮下注射,在上臂,大腿上部,或腹部管理钙和维生素D是必要的治疗或预防低钙血症恶性高钙血症:辖120毫克,每4周增加120毫克剂量的8天15在第一个月的治疗。皮下注射,在上臂,大腿上部,或腹部
狄诺塞麦注射剂英文版说明书
Safety InformationDo not take XGEVA® if you have low blood calcium (hypocalcemia). Your low blood calcium must be treated before you receive XGEVA®. XGEVA® can significantly lower the calcium levels in your blood and some deaths have been reported. Take calcium and vitamin D as your doctor tells you to. Tell your doctor right away if you experience spasms, twitches, cramps, or stiffness in your muscles or numbness or tingling in your fingers, toes, or around your mouth.Do not take XGEVA® if you are allergic to denosumab or any of the ingredients of XGEVA®. Serious allergic reactions have happened in people who take XGEVA®. Call your doctor or go to your nearest emergency room right away if you have any symptoms of a serious allergic reaction, including low blood pressure (hypotension); trouble breathing; throat tightness; swelling of the face, lips, or tongue, rash; itching; or hives.What is the most important information you should know about XGEVA®?Do not take XGEVA® if you take Prolia. XGEVA® contains the same medicine as Prolia® (denosumab).Severe jaw bone problems (osteonecrosis)Severe jaw bone problems may happen when you take XGEVA®. Your doctor should examine your mouth before you start, and while you are taking XGEVA®. Tell your dentist that you are taking XGEVA®. It is important for you to practice good mouth care during treatment with XGEVA®. In studies of patients with bone involvement, the rate of severe jaw problems was higher the longer they were being treated with XGEVA®.Unusual thigh bone fractureUnusual thigh bone fracture has been reported. Symptoms of a fracture include new or unusual pain in your hip, groin, or thigh.Risk of high calcium levels in patients with Giant Cell Tumor of Bone and in patients who are still growingPatients with a type of cancer called Giant Cell Tumor of Bone and patients with bones that are not fully matured are at a greater risk to develop high blood calcium levels after they stop taking XGEVA®, that can be serious.Increased risk of broken bones in the spine after discontinuing XGEVA®After your treatment with XGEVA® is stopped, your risk for breaking bones in your spine can increase, especially if you have a history of risk factors such as osteoporosis or prior fractures.Possible harm to your unborn babyYou should not become pregnant while taking XGEVA®. Tell your doctor right away if you are pregnant, plan to become pregnant, or suspect you are pregnant. XGEVA® can harm your unborn baby.Tell your doctor if you:Are taking a medicine called Prolia® (denosumab) because it contains the same medicine as XGEVA®Have symptoms of low blood calcium such as muscle stiffness or crampsHave symptoms of severe jaw bone problems such as pain or numbnessHave ongoing pain or slow healing after dental surgeryHave symptoms of high blood calcium such as nausea, vomiting, headache, and decreased alertnessAre pregnant, plan to become pregnant, suspect you are pregnant, or breastfeedingWhile taking XGEVA®, you should:Take good care of your teeth and gums and visit a dentist as recommendedTell your dentist that you are taking XGEVA®Tell your doctor if you plan to have dental surgery or teeth removedTalk to your doctor before you stop taking XGEVA® about your risk for broken bones in your spineWomen of child bearing age should use highly effective contraception while taking XGEVA® and for at least 5 months after the last dose of XGEVA®What are the possible side effects of XGEVA®?In patients with bone metastases from solid tumors using XGEVA®, the most common side effects were tiredness/weakness, low phosphate levels in your blood, and nausea. The most common serious side effect of XGEVA® was shortness of breath.In multiple myeloma patients receiving XGEVA®, the most common side effects were diarrhea, nausea, low red blood cells, low blood platelets and calcium levels, back pain, swelling of the lower legs or hands, upper respiratory tract infection, rash, and headache. The most common serious adverse reaction in multiple myeloma patients was pneumonia.These are not all the possible side effects of XGEVA®. For more information, ask your doctor or pharmacist.Call your doctor for medical advice about side effects. You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.Please see Full Prescribing Information.IndicationXGEVA® is a prescription medicine used to prevent fracture, spinal cord compression, or the need for radiation or surgery to bone in patients with multiple myeloma and in patients with bone metastases from solid tumors.
 狄诺塞麦注射剂Xgeva Injektionslösung 120mg/1.7ml[/caption]
药店国别:
产地国家: 瑞士
处方药:是
所属类别: 120毫克/1.7毫升/瓶
包装规格:120毫克/1.7毫升/瓶
计价单位:瓶
生产厂家中文参考译名:安进瑞士AG
生产厂家英文名:Amgen Switzerland AG
原产地英文商品名:Xgeva Inj Loes 120mg/1.7mL/Vial
原产地英文药品名:denosumab
中文参考商品译名:Xgeva注射剂 120毫克/1.7毫升/瓶
中文参考药品译名:Xgeva注射剂 120毫克/1.7毫升/瓶
曾用名:
简介:
部份中文狄迪诺赛麦处方资料(仅供参考)英文药名:Xgeva英文名称:denosumab中文药名:狄迪诺赛麦注射剂药品介绍:Xgeva(狄迪诺塞麦)是一种RANK配体的抑制剂,由Amgen公司生产,2010年11月18日批准用于防止已扩散到骨的癌症,2013年6月13日批准用于治疗骨巨细胞瘤(GCTB)成年和青少年,一种罕见和通常非癌性肿瘤。
适应症:
对有实体瘤且转移的患者预防,狄迪诺赛麦不适合用于多发性骨髓瘤的患者为预防骨骼做预防。
用法用量
Xgeva仅供皮下途径而不应被静脉内,肌内,或皮内骨转移从实体瘤:管理120毫克每4周作为皮下注射在上臂,大腿上部,或腹部骨巨细胞瘤:辖120毫克,每4周增加120毫克剂量的8天15在第一个月的治疗。皮下注射,在上臂,大腿上部,或腹部管理钙和维生素D是必要的治疗或预防低钙血症恶性高钙血症:辖120毫克,每4周增加120毫克剂量的8天15在第一个月的治疗。皮下注射,在上臂,大腿上部,或腹部
狄诺塞麦注射剂英文版说明书
Safety InformationDo not take XGEVA® if you have low blood calcium (hypocalcemia). Your low blood calcium must be treated before you receive XGEVA®. XGEVA® can significantly lower the calcium levels in your blood and some deaths have been reported. Take calcium and vitamin D as your doctor tells you to. Tell your doctor right away if you experience spasms, twitches, cramps, or stiffness in your muscles or numbness or tingling in your fingers, toes, or around your mouth.Do not take XGEVA® if you are allergic to denosumab or any of the ingredients of XGEVA®. Serious allergic reactions have happened in people who take XGEVA®. Call your doctor or go to your nearest emergency room right away if you have any symptoms of a serious allergic reaction, including low blood pressure (hypotension); trouble breathing; throat tightness; swelling of the face, lips, or tongue, rash; itching; or hives.What is the most important information you should know about XGEVA®?Do not take XGEVA® if you take Prolia. XGEVA® contains the same medicine as Prolia® (denosumab).Severe jaw bone problems (osteonecrosis)Severe jaw bone problems may happen when you take XGEVA®. Your doctor should examine your mouth before you start, and while you are taking XGEVA®. Tell your dentist that you are taking XGEVA®. It is important for you to practice good mouth care during treatment with XGEVA®. In studies of patients with bone involvement, the rate of severe jaw problems was higher the longer they were being treated with XGEVA®.Unusual thigh bone fractureUnusual thigh bone fracture has been reported. Symptoms of a fracture include new or unusual pain in your hip, groin, or thigh.Risk of high calcium levels in patients with Giant Cell Tumor of Bone and in patients who are still growingPatients with a type of cancer called Giant Cell Tumor of Bone and patients with bones that are not fully matured are at a greater risk to develop high blood calcium levels after they stop taking XGEVA®, that can be serious.Increased risk of broken bones in the spine after discontinuing XGEVA®After your treatment with XGEVA® is stopped, your risk for breaking bones in your spine can increase, especially if you have a history of risk factors such as osteoporosis or prior fractures.Possible harm to your unborn babyYou should not become pregnant while taking XGEVA®. Tell your doctor right away if you are pregnant, plan to become pregnant, or suspect you are pregnant. XGEVA® can harm your unborn baby.Tell your doctor if you:Are taking a medicine called Prolia® (denosumab) because it contains the same medicine as XGEVA®Have symptoms of low blood calcium such as muscle stiffness or crampsHave symptoms of severe jaw bone problems such as pain or numbnessHave ongoing pain or slow healing after dental surgeryHave symptoms of high blood calcium such as nausea, vomiting, headache, and decreased alertnessAre pregnant, plan to become pregnant, suspect you are pregnant, or breastfeedingWhile taking XGEVA®, you should:Take good care of your teeth and gums and visit a dentist as recommendedTell your dentist that you are taking XGEVA®Tell your doctor if you plan to have dental surgery or teeth removedTalk to your doctor before you stop taking XGEVA® about your risk for broken bones in your spineWomen of child bearing age should use highly effective contraception while taking XGEVA® and for at least 5 months after the last dose of XGEVA®What are the possible side effects of XGEVA®?In patients with bone metastases from solid tumors using XGEVA®, the most common side effects were tiredness/weakness, low phosphate levels in your blood, and nausea. The most common serious side effect of XGEVA® was shortness of breath.In multiple myeloma patients receiving XGEVA®, the most common side effects were diarrhea, nausea, low red blood cells, low blood platelets and calcium levels, back pain, swelling of the lower legs or hands, upper respiratory tract infection, rash, and headache. The most common serious adverse reaction in multiple myeloma patients was pneumonia.These are not all the possible side effects of XGEVA®. For more information, ask your doctor or pharmacist.Call your doctor for medical advice about side effects. You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.Please see Full Prescribing Information.IndicationXGEVA® is a prescription medicine used to prevent fracture, spinal cord compression, or the need for radiation or surgery to bone in patients with multiple myeloma and in patients with bone metastases from solid tumors.
狄诺塞麦注射剂Xgeva Injektionslösung 120mg/1.7ml[/caption]
药店国别:
产地国家: 瑞士
处方药:是
所属类别: 120毫克/1.7毫升/瓶
包装规格:120毫克/1.7毫升/瓶
计价单位:瓶
生产厂家中文参考译名:安进瑞士AG
生产厂家英文名:Amgen Switzerland AG
原产地英文商品名:Xgeva Inj Loes 120mg/1.7mL/Vial
原产地英文药品名:denosumab
中文参考商品译名:Xgeva注射剂 120毫克/1.7毫升/瓶
中文参考药品译名:Xgeva注射剂 120毫克/1.7毫升/瓶
曾用名:
简介:
部份中文狄迪诺赛麦处方资料(仅供参考)英文药名:Xgeva英文名称:denosumab中文药名:狄迪诺赛麦注射剂药品介绍:Xgeva(狄迪诺塞麦)是一种RANK配体的抑制剂,由Amgen公司生产,2010年11月18日批准用于防止已扩散到骨的癌症,2013年6月13日批准用于治疗骨巨细胞瘤(GCTB)成年和青少年,一种罕见和通常非癌性肿瘤。
适应症:
对有实体瘤且转移的患者预防,狄迪诺赛麦不适合用于多发性骨髓瘤的患者为预防骨骼做预防。
用法用量
Xgeva仅供皮下途径而不应被静脉内,肌内,或皮内骨转移从实体瘤:管理120毫克每4周作为皮下注射在上臂,大腿上部,或腹部骨巨细胞瘤:辖120毫克,每4周增加120毫克剂量的8天15在第一个月的治疗。皮下注射,在上臂,大腿上部,或腹部管理钙和维生素D是必要的治疗或预防低钙血症恶性高钙血症:辖120毫克,每4周增加120毫克剂量的8天15在第一个月的治疗。皮下注射,在上臂,大腿上部,或腹部
狄诺塞麦注射剂英文版说明书
Safety InformationDo not take XGEVA® if you have low blood calcium (hypocalcemia). Your low blood calcium must be treated before you receive XGEVA®. XGEVA® can significantly lower the calcium levels in your blood and some deaths have been reported. Take calcium and vitamin D as your doctor tells you to. Tell your doctor right away if you experience spasms, twitches, cramps, or stiffness in your muscles or numbness or tingling in your fingers, toes, or around your mouth.Do not take XGEVA® if you are allergic to denosumab or any of the ingredients of XGEVA®. Serious allergic reactions have happened in people who take XGEVA®. Call your doctor or go to your nearest emergency room right away if you have any symptoms of a serious allergic reaction, including low blood pressure (hypotension); trouble breathing; throat tightness; swelling of the face, lips, or tongue, rash; itching; or hives.What is the most important information you should know about XGEVA®?Do not take XGEVA® if you take Prolia. XGEVA® contains the same medicine as Prolia® (denosumab).Severe jaw bone problems (osteonecrosis)Severe jaw bone problems may happen when you take XGEVA®. Your doctor should examine your mouth before you start, and while you are taking XGEVA®. Tell your dentist that you are taking XGEVA®. It is important for you to practice good mouth care during treatment with XGEVA®. In studies of patients with bone involvement, the rate of severe jaw problems was higher the longer they were being treated with XGEVA®.Unusual thigh bone fractureUnusual thigh bone fracture has been reported. Symptoms of a fracture include new or unusual pain in your hip, groin, or thigh.Risk of high calcium levels in patients with Giant Cell Tumor of Bone and in patients who are still growingPatients with a type of cancer called Giant Cell Tumor of Bone and patients with bones that are not fully matured are at a greater risk to develop high blood calcium levels after they stop taking XGEVA®, that can be serious.Increased risk of broken bones in the spine after discontinuing XGEVA®After your treatment with XGEVA® is stopped, your risk for breaking bones in your spine can increase, especially if you have a history of risk factors such as osteoporosis or prior fractures.Possible harm to your unborn babyYou should not become pregnant while taking XGEVA®. Tell your doctor right away if you are pregnant, plan to become pregnant, or suspect you are pregnant. XGEVA® can harm your unborn baby.Tell your doctor if you:Are taking a medicine called Prolia® (denosumab) because it contains the same medicine as XGEVA®Have symptoms of low blood calcium such as muscle stiffness or crampsHave symptoms of severe jaw bone problems such as pain or numbnessHave ongoing pain or slow healing after dental surgeryHave symptoms of high blood calcium such as nausea, vomiting, headache, and decreased alertnessAre pregnant, plan to become pregnant, suspect you are pregnant, or breastfeedingWhile taking XGEVA®, you should:Take good care of your teeth and gums and visit a dentist as recommendedTell your dentist that you are taking XGEVA®Tell your doctor if you plan to have dental surgery or teeth removedTalk to your doctor before you stop taking XGEVA® about your risk for broken bones in your spineWomen of child bearing age should use highly effective contraception while taking XGEVA® and for at least 5 months after the last dose of XGEVA®What are the possible side effects of XGEVA®?In patients with bone metastases from solid tumors using XGEVA®, the most common side effects were tiredness/weakness, low phosphate levels in your blood, and nausea. The most common serious side effect of XGEVA® was shortness of breath.In multiple myeloma patients receiving XGEVA®, the most common side effects were diarrhea, nausea, low red blood cells, low blood platelets and calcium levels, back pain, swelling of the lower legs or hands, upper respiratory tract infection, rash, and headache. The most common serious adverse reaction in multiple myeloma patients was pneumonia.These are not all the possible side effects of XGEVA®. For more information, ask your doctor or pharmacist.Call your doctor for medical advice about side effects. You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch, or call 1-800-FDA-1088.Please see Full Prescribing Information.IndicationXGEVA® is a prescription medicine used to prevent fracture, spinal cord compression, or the need for radiation or surgery to bone in patients with multiple myeloma and in patients with bone metastases from solid tumors.
用药温馨提示:当您服用此药物时,需定期接受医疗专业人士的检查,以便随时针对其药效、副作用等情况进行监测。本网站所包含的信息旨在为患者提供帮助,不能代替医学建议和治疗。
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 狄诺塞麦注射剂Xgeva Injektionslösung 120mg/1.7ml
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 狄诺塞麦注射剂Xgeva Injektionslösung 120mg/1.7ml