二联复方sofosbuvir/ledipasvir filmcoated tablets(Harvoni)说明书
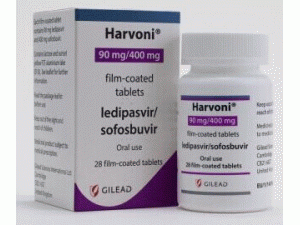 产地国家:英国
处方药:是
所属类别: (90毫克/400毫克)/片 28片/瓶
包装规格: (90毫克/400毫克)/片 28片/瓶
计价单位:瓶
生产厂家英文名:Gilead
原产地英文商品名:Harvoni tab(90mg/400mg)/tab 28tabs/box
原产地英文药品名:sofosbuvir+ledipasvir
中文参考商品译名:Harvoni二联复方薄膜包衣片(90毫克/400毫克)/片 28片/瓶
中文参考药品译名:sofosbuvir+ledipasvir
产地国家:英国
处方药:是
所属类别: (90毫克/400毫克)/片 28片/瓶
包装规格: (90毫克/400毫克)/片 28片/瓶
计价单位:瓶
生产厂家英文名:Gilead
原产地英文商品名:Harvoni tab(90mg/400mg)/tab 28tabs/box
原产地英文药品名:sofosbuvir+ledipasvir
中文参考商品译名:Harvoni二联复方薄膜包衣片(90毫克/400毫克)/片 28片/瓶
中文参考药品译名:sofosbuvir+ledipasvir
简介
Harvoni是第一个被批准治疗慢性HCV基因型1 感染二联复方药丸。第一个被批准不需要干扰素或利巴韦林的方案。 FDA药品评价和研究中心抗微生物产品室主任说:“随着发展和批准对丙型肝炎病毒新治疗,我们正在改变有此病美国人生活的治疗范式,”“直至去年,对丙型肝炎唯一得到的治疗需要给予干扰素和利巴韦林。现患者和卫生保健专业人员有多种治疗选择,包括二联复方药丸有助于简化治疗方案。”Harvoni是在去年治疗以来被批准治疗男性丙型肝炎的第三个药物;第七个接到PDA批准突破性治疗指定的新药。批准日期:10月10,2014年10月10日;公司:Gilead Sciences,IncHARVONITM(ledipasvir和sofosbuvir)片为口服使用美国初次批准:2014 适应证和用途 HARVONI是一个ledipasvir,一种丙肝病毒(HCV)NS5A抑制剂,和sofosbuvir,一种HCV核苷酸类似物NS5B聚合酶抑制剂的固定剂量组合复方,和适用在成年中为慢性丙肝(CHC)基因型1感染的治疗。 剂量和给药方法 ⑴ 推荐剂量:1片(90 mg ledipasvir和400 mg sofosbuvir)口服每天1次有或无食物。⑵ 推荐治疗时间(2.1):⒈未治疗过有或无肝硬化:12周⒉经历治疗无肝硬化:12周⒊经历治疗有肝硬化:24周⑶ 对有严重肾受损或肾病终末期患者不能做推荐剂量剂型和规格片:90 mg ledipasvir和400 mg sofosbuvir。 禁忌证:无 警告和注意事项 不建议与其他含sofosbuvir其他药物使用,包括SOVALDI。 不良反应 用HARVONI治疗共8,12,或24周观察到最常见不良反应(发生率大于或等于10%,所有级别)是疲乏和头痛。 药物相互作用 ⑴ P-gp诱导剂(如,利福平[rifampin],圣约翰草[St. John’s wort]):可能改变ledipasvir和sofosbuvir的浓度。不建议HARVONI与P-gp诱导剂使用。⑵ 使用前为药物相互作用潜在咨询完整处方资料。英文版说明书
U.S. Food and Drug Administration Approves Gilead’s Harvoni® (Ledipasvir/Sofosbuvir), the First Once-Daily Single Tablet Regimen for the Treatment of Genotype 1 Chronic Hepatitis C-- Harvoni Achieved Cure Rates (SVR12) of 94-99 Percent in Three Phase 3 Studies-- Shortens Treatment Duration to Just Eight Weeks for Certain Treatment-Naïve Patients-- Eliminates Need for Interferon and Ribavirin for Patients with Genotype 1 Hepatitis CFOSTER CITY, Calif.--(BUSINESS WIRE)--Oct. 10, 2014-- Gilead Sciences, Inc. (Nasdaq: GILD) today announced that the U.S. Food and Drug Administration (FDA) has approved Harvoni® (ledipasvir 90 mg/sofosbuvir 400 mg), the first once-daily single tablet regimen for the treatment of chronic hepatitis C genotype 1 infection in adults. Harvoni combines the NS5A inhibitor ledipasvir with the nucleotide analog polymerase inhibitor sofosbuvir, approved under the tradename Sovaldi® in December 2013. Harvoni’s efficacy has been established in patients with chronic hepatitis C virus (HCV) genotype 1 infection, with a treatment duration of eight, 12 or 24 weeks depending on prior treatment history, cirrhosis status and baseline viral load. Eight weeks of treatment with Harvoni can be considered for treatment-naïve patients without cirrhosis who have baseline HCV viral load below 6 million IU/mL.The FDA granted Harvoni a Priority Review and Breakthrough Therapy designation, which is given to investigational medicines that may offer major advances in treatment over available therapies.“By providing very high cure rates in as little as eight weeks and completely eliminating the need for interferon and ribavirin, which are challenging to take and tolerate, Harvoni significantly advances treatment for patients with the most common form of hepatitis C in the United States,” said Nezam Afdhal, MD, Director of Hepatology at Beth Israel Deaconess Medical Center, Professor of Medicine at Harvard Medical School and a principal investigator in the Harvoni clinical trials. “For the first time, the vast majority of patients can be cured with a once-daily pill in only eight or 12 weeks.”Harvoni’s approval is supported by data from three Phase 3 studies, ION-1, ION-2 and ION-3. These studies eva luated eight, 12 or 24 weeks of treatment with Harvoni, with or without ribavirin, among nearly 2,000 genotype 1 HCV patients with compensated liver disease. These studies included non-cirrhotic treatment-naïve patients (ION-3), cirrhotic and non-cirrhotic treatment-naïve patients (ION-1) and cirrhotic and non-cirrhotic patients who failed prior therapy with an interferon-based regimen, including regimens containing an HCV protease inhibitor (ION-2). The primary endpoint for each study was sustained virologic response (HCV undetectable) 12 weeks after completing therapy (SVR12).Patients who achieve SVR12 are considered cured of HCV. In these studies, ribavirin was not shown to increase response rates. Trial participants in the ribavirin-free arms (n=863) achieved SVR12 rates of 94 to 99 percent.“Unlike other serious chronic diseases, hepatitis C can be cured and Harvoni offers patients the potential for a cure in as little as eight weeks,” said John C. Martin, PhD, Chairman and Chief Executive Officer, Gilead Sciences. “Gilead is proud to have played a role in developing a once-daily therapy that is safe, simple and well tolerated. We are now working to ensure rapid and broad access to Harvoni.”Important Safety Information regarding warnings and precautions, adverse reactions and drug interactions is listed below. Zero percent, less than 1 percent and 1 percent of patients treated for eight, 12 and 24 weeks, respectively, discontinued treatment due to adverse events and fewer adverse events were observed in the ribavirin-free arms compared to the ribavirin-containing arms in all ION studies. The most common adverse reactions among patients treated with Harvoni (≥5 percent) were fatigue, headache, nausea, diarrhea and insomnia. For additional study details, and complete dosing information, see the Clinical Studies and Dosage and Administration sections, respectively, of the full Prescribing Information.IMPORTANT SAFETY INFORMATIONWarnings and PrecautionsRisk of Reduced Therapeutic Effect of Harvoni Due to P-gp Inducers: Rifampin and St. John’s wort are not recommended for use with Harvoni as they may significantly decrease ledipasvir and sofosbuvir plasma concentrations.Related Products Not Recommended: Harvoni is not recommended for use with other products containing sofosbuvir (Sovaldi).Adverse ReactionsMost common (≥10%, all grades) adverse reactions were fatigue and headache.Drug InteractionsIn addition to rifampin and St. John’s wort, coadministration of Harvoni is also not recommended with carbamazepine, oxcarbazepine, phenobarbital, phenytoin, rifabutin, rifapentine, and tipranavir/ritonavir. Such coadministration is expected to decrease the concentration of ledipasvir and sofosbuvir, reducing the therapeutic effect of Harvoni.Coadministration of Harvoni is not recommended with simeprevir due to increased concentrations of ledipasvir and simeprevir. Coadministration is also not recommended with rosuvastatin or co-formulated elvitegravir/cobicistat/emtricitabine/tenofovir disoproxil fumarate due to increased concentrations of rosuvastatin and tenofovir, respectively.Consult the full Prescribing Information for Harvoni for more information on potentially significant drug interactions, including clinical comments.About Gilead SciencesGilead Sciences is a biopharmaceutical company that discovers, develops and commercializes innovative therapeutics in areas of unmet medical need. The company’s mission is to advance the care of patients suffering from life-threatening diseases worldwide. Headquartered in Foster City, California, Gilead has operations in North and South America, Europe and Asia Pacific.用药温馨提示:当您服用此药物时,需定期接受医疗专业人士的检查,以便随时针对其药效、副作用等情况进行监测。本网站所包含的信息旨在为患者提供帮助,不能代替医学建议和治疗。
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 二联复方sofosbuvir/ledipasvir filmcoated tablets(Harvoni)说明书
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 二联复方sofosbuvir/ledipasvir filmcoated tablets(Harvoni)说明书