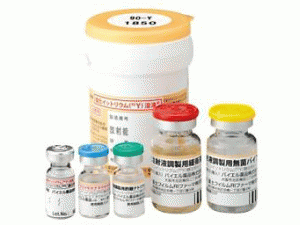
替伊莫单抗Zeva lin yttrium injection kit Y-90
 药店国别:
产地国家:日本
处方药:是
所属类别: Y-90 1套
包装规格: Y-90 1套
计价单位:套
生产厂家中文参考译名:
生产厂家英文名:Spectra Pharmaceutical
原产地英文商品名:Zeva lin yttrium injection(ゼヴァリンイットリウム(90Y)静注用セット)Y-90 1kit
原产地英文药品名:Ibritumomab Tiuxetan(genetical recombination)
中文参考商品译名:泽娃灵(ゼヴァリンイットリウム(90Y)静注用セット)Y-90 1套
中文参考药品译名:替伊莫单抗/人血白蛋白
曾用名:
简介:
部份中文替伊莫单抗处方资料(仅供参考)英文名:Ibritumomab Tiuxetan商品名:Zeva lIN中文名:替伊莫单抗注射剂生产商:光谱制药ゼヴァリン イットリウム(90Y)静注用セット药用类别名称抗肿瘤药物与放射性标记抗CD20单克隆抗体批准日期:2008年6月商標名Zeva lIN yttrium injection一般名イブリツモマブ チウキセタン(遺伝子組換え)Ibritumomab Tiuxetan(genetical recombination)〔JAN〕本質中国仓鼠卵巢细胞中产生的213个氨基酸残基是一种小鼠抗人类CD20单克隆抗体 (c1018h15615n276o333s7)。
分子量:23, 221.42)光链2个分子, 由445氨基酸残基组成 (C2183H3334 N564O671S21; 分子量:48, 888.57)糖蛋白由n-{(2S)-2-[之二(羧甲基) 氨基]-3-(4-异硫氰酸酯-苯基) 丙基}-n {2-bis(羧甲基)丙基} 氨基(C23H30N4O10) 组成的重链2分子组成。S; 分子量: 554. 57)是一种结合在一起的改性糖蛋白(重组)。分子量148kDa(道尔顿)放射性核素的特性90Y理化半衰期64.1小时Β射线能量2.281 MeV(99.98)减衰表経過時間(時間)残存放射能(%)経過時間(時間)残存放射能(%)経過時間(時間)残存放射能
批准条件由于日本的临床试验病例极为有限, 在生产和销售之后, 直到积累了一定数量病例的数据, 通过对所有病例进行使用结果调查, 对所有病例进行了(90Y)伊布里佐穆马 尽早掌握使用Tiuxetan(转基因) 患者的背景信息, 并收集有关(90Y)的安全性和有效性的数据, 早期, (90Y) 采取必要措施, 正确使用替克西坦 (转基因)。药
用药理学
1. cd20抗原人类CD20抗原是分化抗原(磷蛋白), 除亲b细胞和血浆细胞外, 几乎所有正常和致瘤b 细胞都表达, 在B细胞以外的细胞中不表达。
2. 抗肿瘤作用在与拉莫斯肿瘤细胞移植的小鼠肿瘤模型中, 观察到(90Y)伊布朱穆纳布-蒂奥克萨坦(重组) 具有肿瘤作用(体内)。
3. 作用机制伊布里佐穆单抗对B细胞上的CD20抗原具有较强的抗原特异性结合能力。作为一种螯合剂, Tiuxetan(与90Y 有强烈的结合), 在抗体中共价地与暴露在外的赖氨酸氨基和精氨酸结合。Yttrium(90Y)ibritzumab Tiuxetan(重组)与CD20抗原以及利妥昔单抗(重组) 结合, 诱导90Y 细胞凋亡和β射线释放, 诱导细胞毒性。
适应症
CD20阳性复发或难治性继发疾病低级b细胞非霍奇金淋巴瘤、地幔细胞淋巴瘤
用法与用量
注射溶液制备用于该组的无菌小瓶制备注射溶液的醋酸钠溶液的适当量,并把氯化钇(90Y)溶液1500MBq,这是通过加入替伊莫单抗溶液1.3毫升,注射溶液制备的适当量的缓冲液混合此外,该解决方案是钇(90Y)替伊莫单抗(遗传重组)注射。 (请参见“关于申请注意事项”)通常情况下,成人,利妥昔单抗(基因重组)静脉输注后,迅速,静脉给药超过14.8MBq/公斤(最高1184MBq)步行10分钟钇(90Y)替伊莫单抗(基因重组) 。另外,在11.1MBq/公斤的减少,这取决于患者的状况。另外,钇(90Y)替伊莫单抗(遗传重组)之前,注射溶液的施用,并且确认替伊莫单抗(遗传重组)的整合位点,以检查异常的生物分布。
包装
泽娃灵钇(90Y),用于静脉注射设定1套
制造商
(进口)光谱制药有限责任公司
英文版说明书
Company: Biogen IDECApproval Status: Approved February 2002Treatment for: Non-Hodgkin's lymphomaAreas: OncologyGeneral InformationIn February 2002, Zeva lin was the first radioimmunotherapy to receive FDA approval. Zeva lin is indicated for the treatment of relapsed or refractory low grade, follicular, or transformed B-cell non-Hodgkin's lymphoma (NHL). This indication includes patients with Rituxan (rituximab)-refractory follicular NHL. Zeva lin has been approved as part of a therapeutic regimen involving Rituxan.Zeva lin consists of a monoclonal antibody linked to the radioactive isotope yttrium-90. After infusion into a patient, the monoclonal antibody targets the CD20 antigen, which is found on the surface of mature B cells and B-cell tumors. In this manner, cytotoxic radiation is delivered directly to malignant cells.Clinical ResultsZeva lin received both a full approval and an accelerated approval based on results from two major US efficacy studies.The study that supported the full approval of Zeva lin included 54 subjects.The subjects were diagnosed with relapsed follicular lymphoma, and they no longer adequately responded to Rituxan treatment. An overall response rate of 74% was achieved with Zeva lin treatment, with 15% of subjects experiencing a complete response.Accelerated approval of Zeva lin was supported by a randomized, controlled phase III trial.The trial included 143 subjects with relapsed or refractory, low grade or follicular NHL or transformed B-cell NHL.An overall response rate of 80% was obtained in subjects receiving the Zeva lin therapeutic regimen (73 subjects), compared to 56% for the subjects receiving Rituxan alone (70 subjects). Thirty percent of Zeva lin-treated subjects experienced a complete response, compared to a 16% complete response rate for Rituxan-treatedsubjects.Side EffectsIn clinical trials, serious adverse reactions caused by the Zeva lin therapeutic regimen included infections, allergic reactions, and hemorrhage while thrombocytopenic. In addition, the development of myeloid malignancies and dysplasias have been reported.Adverse events that occurred in greater than 5% of subjects include(but are not limited to) the following:NauseaVomitingDiarrheaAnorexiaThrombocytopenia (decreased number of blood platelets)Neutropenia (decreased number of white blood cells)AnemiaArthralgia(joint pain)DizzinessDyspnea(difficult or labored breathing)Increased coughMechanism of ActionThe complementarity-determining regions of Ibritumomab bind to the CD20 antigen on B lymphocytes.Ibritumomab, like Rituximab, induces apoptosis in CD20+ B-cell lines in vitro. The chelate tiuxetan, which tightly binds In-111 or Y-90, is covalently linked to theamino groups of exposed lysines and arginines contained within the antibody.The beta emission from Y-90 induces cellular damage by the formation of free radicals in the target and neighboring cells.(from Zeva lin Prescribing Information)Additional InformationSchering AG has the marketing rights to Zeva lin outside of the United States.For more information on Zeva lin, please visit IDEC Pharmaceuticals.Additional information on non-Hodgkin's lymphoma can be obtained through the National Cancer Institute.
药店国别:
产地国家:日本
处方药:是
所属类别: Y-90 1套
包装规格: Y-90 1套
计价单位:套
生产厂家中文参考译名:
生产厂家英文名:Spectra Pharmaceutical
原产地英文商品名:Zeva lin yttrium injection(ゼヴァリンイットリウム(90Y)静注用セット)Y-90 1kit
原产地英文药品名:Ibritumomab Tiuxetan(genetical recombination)
中文参考商品译名:泽娃灵(ゼヴァリンイットリウム(90Y)静注用セット)Y-90 1套
中文参考药品译名:替伊莫单抗/人血白蛋白
曾用名:
简介:
部份中文替伊莫单抗处方资料(仅供参考)英文名:Ibritumomab Tiuxetan商品名:Zeva lIN中文名:替伊莫单抗注射剂生产商:光谱制药ゼヴァリン イットリウム(90Y)静注用セット药用类别名称抗肿瘤药物与放射性标记抗CD20单克隆抗体批准日期:2008年6月商標名Zeva lIN yttrium injection一般名イブリツモマブ チウキセタン(遺伝子組換え)Ibritumomab Tiuxetan(genetical recombination)〔JAN〕本質中国仓鼠卵巢细胞中产生的213个氨基酸残基是一种小鼠抗人类CD20单克隆抗体 (c1018h15615n276o333s7)。
分子量:23, 221.42)光链2个分子, 由445氨基酸残基组成 (C2183H3334 N564O671S21; 分子量:48, 888.57)糖蛋白由n-{(2S)-2-[之二(羧甲基) 氨基]-3-(4-异硫氰酸酯-苯基) 丙基}-n {2-bis(羧甲基)丙基} 氨基(C23H30N4O10) 组成的重链2分子组成。S; 分子量: 554. 57)是一种结合在一起的改性糖蛋白(重组)。分子量148kDa(道尔顿)放射性核素的特性90Y理化半衰期64.1小时Β射线能量2.281 MeV(99.98)减衰表経過時間(時間)残存放射能(%)経過時間(時間)残存放射能(%)経過時間(時間)残存放射能
批准条件由于日本的临床试验病例极为有限, 在生产和销售之后, 直到积累了一定数量病例的数据, 通过对所有病例进行使用结果调查, 对所有病例进行了(90Y)伊布里佐穆马 尽早掌握使用Tiuxetan(转基因) 患者的背景信息, 并收集有关(90Y)的安全性和有效性的数据, 早期, (90Y) 采取必要措施, 正确使用替克西坦 (转基因)。药
用药理学
1. cd20抗原人类CD20抗原是分化抗原(磷蛋白), 除亲b细胞和血浆细胞外, 几乎所有正常和致瘤b 细胞都表达, 在B细胞以外的细胞中不表达。
2. 抗肿瘤作用在与拉莫斯肿瘤细胞移植的小鼠肿瘤模型中, 观察到(90Y)伊布朱穆纳布-蒂奥克萨坦(重组) 具有肿瘤作用(体内)。
3. 作用机制伊布里佐穆单抗对B细胞上的CD20抗原具有较强的抗原特异性结合能力。作为一种螯合剂, Tiuxetan(与90Y 有强烈的结合), 在抗体中共价地与暴露在外的赖氨酸氨基和精氨酸结合。Yttrium(90Y)ibritzumab Tiuxetan(重组)与CD20抗原以及利妥昔单抗(重组) 结合, 诱导90Y 细胞凋亡和β射线释放, 诱导细胞毒性。
适应症
CD20阳性复发或难治性继发疾病低级b细胞非霍奇金淋巴瘤、地幔细胞淋巴瘤
用法与用量
注射溶液制备用于该组的无菌小瓶制备注射溶液的醋酸钠溶液的适当量,并把氯化钇(90Y)溶液1500MBq,这是通过加入替伊莫单抗溶液1.3毫升,注射溶液制备的适当量的缓冲液混合此外,该解决方案是钇(90Y)替伊莫单抗(遗传重组)注射。 (请参见“关于申请注意事项”)通常情况下,成人,利妥昔单抗(基因重组)静脉输注后,迅速,静脉给药超过14.8MBq/公斤(最高1184MBq)步行10分钟钇(90Y)替伊莫单抗(基因重组) 。另外,在11.1MBq/公斤的减少,这取决于患者的状况。另外,钇(90Y)替伊莫单抗(遗传重组)之前,注射溶液的施用,并且确认替伊莫单抗(遗传重组)的整合位点,以检查异常的生物分布。
包装
泽娃灵钇(90Y),用于静脉注射设定1套
制造商
(进口)光谱制药有限责任公司
英文版说明书
Company: Biogen IDECApproval Status: Approved February 2002Treatment for: Non-Hodgkin's lymphomaAreas: OncologyGeneral InformationIn February 2002, Zeva lin was the first radioimmunotherapy to receive FDA approval. Zeva lin is indicated for the treatment of relapsed or refractory low grade, follicular, or transformed B-cell non-Hodgkin's lymphoma (NHL). This indication includes patients with Rituxan (rituximab)-refractory follicular NHL. Zeva lin has been approved as part of a therapeutic regimen involving Rituxan.Zeva lin consists of a monoclonal antibody linked to the radioactive isotope yttrium-90. After infusion into a patient, the monoclonal antibody targets the CD20 antigen, which is found on the surface of mature B cells and B-cell tumors. In this manner, cytotoxic radiation is delivered directly to malignant cells.Clinical ResultsZeva lin received both a full approval and an accelerated approval based on results from two major US efficacy studies.The study that supported the full approval of Zeva lin included 54 subjects.The subjects were diagnosed with relapsed follicular lymphoma, and they no longer adequately responded to Rituxan treatment. An overall response rate of 74% was achieved with Zeva lin treatment, with 15% of subjects experiencing a complete response.Accelerated approval of Zeva lin was supported by a randomized, controlled phase III trial.The trial included 143 subjects with relapsed or refractory, low grade or follicular NHL or transformed B-cell NHL.An overall response rate of 80% was obtained in subjects receiving the Zeva lin therapeutic regimen (73 subjects), compared to 56% for the subjects receiving Rituxan alone (70 subjects). Thirty percent of Zeva lin-treated subjects experienced a complete response, compared to a 16% complete response rate for Rituxan-treatedsubjects.Side EffectsIn clinical trials, serious adverse reactions caused by the Zeva lin therapeutic regimen included infections, allergic reactions, and hemorrhage while thrombocytopenic. In addition, the development of myeloid malignancies and dysplasias have been reported.Adverse events that occurred in greater than 5% of subjects include(but are not limited to) the following:NauseaVomitingDiarrheaAnorexiaThrombocytopenia (decreased number of blood platelets)Neutropenia (decreased number of white blood cells)AnemiaArthralgia(joint pain)DizzinessDyspnea(difficult or labored breathing)Increased coughMechanism of ActionThe complementarity-determining regions of Ibritumomab bind to the CD20 antigen on B lymphocytes.Ibritumomab, like Rituximab, induces apoptosis in CD20+ B-cell lines in vitro. The chelate tiuxetan, which tightly binds In-111 or Y-90, is covalently linked to theamino groups of exposed lysines and arginines contained within the antibody.The beta emission from Y-90 induces cellular damage by the formation of free radicals in the target and neighboring cells.(from Zeva lin Prescribing Information)Additional InformationSchering AG has the marketing rights to Zeva lin outside of the United States.For more information on Zeva lin, please visit IDEC Pharmaceuticals.Additional information on non-Hodgkin's lymphoma can be obtained through the National Cancer Institute.
用药温馨提示:当您服用此药物时,需定期接受医疗专业人士的检查,以便随时针对其药效、副作用等情况进行监测。本网站所包含的信息旨在为患者提供帮助,不能代替医学建议和治疗。
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 替伊莫单抗Zeva lin yttrium injection kit Y-90
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 替伊莫单抗Zeva lin yttrium injection kit Y-90