曲氟尿苷盐酸/替吡嘧啶复方片Lonsurf Tablets 15/6.14mg说明书
 药店国别:
产地国家:美国
处方药:是
所属类别: 15毫克/6.14毫克/片 40片/瓶
包装规格: 15毫克/6.14毫克/片 40片/瓶
计价单位:瓶
生产厂家中文参考译名:
生产厂家英文名:TAIHO ONCOLOGY INC
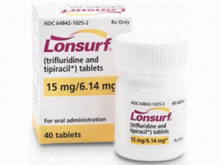
原产地英文商品名:LONSURF 15/6.14mg/Tablets 40Tablets/bottles
原产地英文药品名:trifluridine and tipiracil
中文参考商品译名:LONSURF复方片 15毫克/6.14毫克/片 40片/瓶
中文参考药品译名:曲氟尿苷盐酸/替吡嘧啶
曾用名:
简介:
近日,美国食品和药物管理局(FDA)已批准Lonsurf(trifluridine/tipiracil,TAS-102)用于既往已接受至少2种化疗方案(包括氟嘧啶、铂,或紫杉烷或伊立替康;如果适用,HER2/neu靶向疗法)的转移性胃或胃食管交界腺癌成人患者的治疗。此次批准,扩大了Lonsurf在美国的适应症,该药之前已获批用于既往已接受标准化疗的转移性结直肠癌(mCRC)患者。批准日期:2015年9月22日;公司:Taiho Oncology,Inc.LONSURF(曲氟尿苷盐酸/替吡嘧啶薄膜片[trifluridine and tipiracil])片剂,用于口服美国最初批准:2015年
作用机制
LONSURF由基于胸苷的核苷类似物,三氟尿苷和胸苷磷酸化酶抑制剂,tipiracil组成,摩尔比为1:0.5(重量比,1:0.471)。包含ofpipiilil通过胸苷磷酸化酶抑制其代谢来增加三氟尿苷的暴露。在摄取到癌细胞中后,将三氟尿苷掺入DNA中,干扰DNA合成并抑制细胞增殖。三氟尿苷/tipiracil在小鼠中表现出对抗KRAS野生型和突变型人结直肠癌异种移植物的抗肿瘤活性。
适应症和用法
LONSURF是一种三氟尿苷(一种核苷代谢抑制剂)和tipiracil(一种胸苷磷酸化酶抑制剂)的组合,用于治疗成年患者:•转移性结直肠癌,之前曾接受氟嘧啶,奥沙利铂和伊立替康为基础的化疗,抗VEGF生物治疗,以及RAS野生型,抗EGFR治疗。•转移性胃或胃食管连接腺癌,至少用两种化疗方案进行治疗,包括氟嘧啶,铂,紫杉烷或伊立替康,以及HER2/neu靶向治疗。
剂量和给药
•推荐剂量:每天两次口服35mg/m2/剂,每天28天的第1天至第5天和第8天至第12天食用。
剂量形式和强度
片剂:•15mg三氟尿苷/6.14mg tipiracil•20mg三氟尿苷/8.19mg tipiracil禁忌症没有。
警告和注意事项
•严重的骨髓抑制:在每个周期的第15天之前和之后获得全血计数。根据建议扣留并恢复下一个较低的LONSURFdosage。•胚胎 - 胎儿毒性:可能导致胎儿伤害。告知女性对胎儿潜在风险的生殖潜力并使用有效的感染。
不良反应
最常见的不良反应或实验室异常(≥10%)区域性贫血,中性粒细胞减少,疲劳/虚弱,恶心,血小板减少,萎缩,腹泻,呕吐和发热。
用于特定人群
•哺乳期:建议不要母乳喂养。•老年人使用:3岁或4岁的中性粒细胞减少症和血小板减少症以及第3级贫血症在65岁或以上的患者中更常见。•肝功能损害:不要在基线中度或严重肝功能损害的患者中启动LONSURF。
包装提供/存储和处理
LONSURF 15mg/6.14mg片剂以白色,双凸面,圆形,薄膜包衣片剂形式提供,一面印有'15',另一面印有'102'和'15mg',灰色墨水。在以下介绍中,这些药片采用HDPE瓶包装,并带有儿童防护罩:•20计数:NDC 64842-1025-1•40计数:NDC 64842-1025-2•60计数:NDC 64842-1025-3LONSURF 20mg/8.19mg片剂以浅红色,双凸面,圆形薄膜包衣片剂形式提供,一面印有'20',另一面印有'102'和'20mg',灰色墨水。在以下介绍中,这些药片采用HDPE瓶包装,并带有儿童防护罩:•20计数:NDC 64842-1020-1•40计数:NDC 64842-1020-2•60计数:NDC 64842-1020-3储存在20°C至25°C(68°F至77°F);允许从15°C到30°C(59°F到50°C)的偏移86°F)[见USP受控室温]。LONSURF是一种细胞毒性药物。遵循适用的特殊处理和处理程序.1如果存放在原瓶外,30天后丢弃。
英文版说明书
SAFETY INFORMATIONWARNINGS AND PRECAUTIONSSevere Myelosuppression: LONSURF caused severe and life‑threatening myelosuppression (Grade 3‑4) consisting of neutropenia (38%), anemia (18%), thrombocytopenia (5%), and febrile neutropenia (3%). Two patients (0.2%) died due to neutropenic infection. A total of 12% of LONSURF‑treated patients received granulocyte‑colony stimulating factors. Obtain complete blood counts prior to and on day 15 of each cycle of LONSURF and more frequently as clinically indicated. Withhold LONSURF for febrile neutropenia, absolute neutrophil count less than 500/mm3, or platelets less than 50,000/mm3. Upon recovery, resume LONSURF at a reduced dose as clinically indicated.Embryo‑Fetal Toxicity: LONSURF can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment and for at least 6 months after the final dose.USE IN SPECIFIC POPULATIONSLactation: It is not known whether LONSURF or its metabolites are present in human milk. There are no data to assess the effects of LONSURF or its metabolites on the breast‑fed infant or the effects on milk production. Because of the potential for serious adverse reactions in breast‑fed infants, advise women not to breastfeed during treatment with LONSURF and for 1 day following the final dose.Male Contraception: Because of the potential for genotoxicity, advise males with female partners of reproductive potential to use condoms during treatment with LONSURF and for at least 3 months after the final dose.Geriatric Use: Patients 65 years of age or over who received LONSURF had a higher incidence of the following compared to patients younger than 65 years: Grade 3 or 4 neutropenia (46% vs 32%), Grade 3 anemia (22% vs 16%), and Grade 3 or 4 thrombocytopenia (7% vs 4%).Hepatic Impairment: Do not initiate LONSURF in patients with baseline moderate or severe (total bilirubin greater than 1.5 times ULN and any AST) hepatic impairment. Patients with severe hepatic impairment (total bilirubin greater than 3 times ULN and any AST) were not studied. No adjustment to the starting dose of LONSURF is recommended for patients with mild hepatic impairment.Renal Impairment: No adjustment to the starting dosage of LONSURF is recommended in patients with mild or moderate renal impairment (CLcr of 30 to 89 mL/min). Patients with severe renal impairment (CLcr < 30 mL/min) were not studied.ADVERSE REACTIONSMost Common Adverse Drug Reactions in Patients Treated With LONSURF (≥5%): The most common adverse drug reactions in LONSURF‑treated patients vs placebo‑treated patients with mCRC, respectively, were asthenia/fatigue (52% vs 35%), nausea (48% vs 24%), decreased appetite (39% vs 29%), diarrhea (32% vs 12%), vomiting (28% vs 14%), infections (27% vs 16%), abdominal pain (21% vs 18%), pyrexia (19% vs 14%), stomatitis (8% vs 6%), dysgeusia (7% vs 2%), and alopecia (7% vs 1%). In metastatic gastric cancer or gastroesophageal junction (GEJ), the most common adverse drug reactions respectively were, nausea (37% vs 32%), decreased appetite (34% vs 31%), vomiting (25% vs 20%), infections (23% vs 16%) and diarrhea (23% vs 14%).Pulmonary emboli occurred more frequently in LONSURF‑treated patients compared to placebo: (2% vs 0%) in mCRC and (3% vs 2%) in metastatic gastric cancer and GEJ.Interstitial lung disease (0.2%), including fatalities, has been reported in clinical studies and clinical practice settings in Asia.Laboratory Test Abnormalities in Patients Treated With LONSURF: Laboratory test abnormalities in LONSURF‑treated patients vs placebo‑treated patients with mCRC, respectively, were anemia (77% vs 33%), neutropenia (67% vs 1%), and thrombocytopenia (42% vs 8%). In metastatic gastric cancer or GEJ, the test abnormalities, respectively, were neutropenia (66% vs 4%), anemia (63% vs 38%), and thrombocytopenia (34% vs 9%).
药店国别:
产地国家:美国
处方药:是
所属类别: 15毫克/6.14毫克/片 40片/瓶
包装规格: 15毫克/6.14毫克/片 40片/瓶
计价单位:瓶
生产厂家中文参考译名:
生产厂家英文名:TAIHO ONCOLOGY INC
原产地英文商品名:LONSURF 15/6.14mg/Tablets 40Tablets/bottles
原产地英文药品名:trifluridine and tipiracil
中文参考商品译名:LONSURF复方片 15毫克/6.14毫克/片 40片/瓶
中文参考药品译名:曲氟尿苷盐酸/替吡嘧啶
曾用名:
简介:
近日,美国食品和药物管理局(FDA)已批准Lonsurf(trifluridine/tipiracil,TAS-102)用于既往已接受至少2种化疗方案(包括氟嘧啶、铂,或紫杉烷或伊立替康;如果适用,HER2/neu靶向疗法)的转移性胃或胃食管交界腺癌成人患者的治疗。此次批准,扩大了Lonsurf在美国的适应症,该药之前已获批用于既往已接受标准化疗的转移性结直肠癌(mCRC)患者。批准日期:2015年9月22日;公司:Taiho Oncology,Inc.LONSURF(曲氟尿苷盐酸/替吡嘧啶薄膜片[trifluridine and tipiracil])片剂,用于口服美国最初批准:2015年
作用机制
LONSURF由基于胸苷的核苷类似物,三氟尿苷和胸苷磷酸化酶抑制剂,tipiracil组成,摩尔比为1:0.5(重量比,1:0.471)。包含ofpipiilil通过胸苷磷酸化酶抑制其代谢来增加三氟尿苷的暴露。在摄取到癌细胞中后,将三氟尿苷掺入DNA中,干扰DNA合成并抑制细胞增殖。三氟尿苷/tipiracil在小鼠中表现出对抗KRAS野生型和突变型人结直肠癌异种移植物的抗肿瘤活性。
适应症和用法
LONSURF是一种三氟尿苷(一种核苷代谢抑制剂)和tipiracil(一种胸苷磷酸化酶抑制剂)的组合,用于治疗成年患者:•转移性结直肠癌,之前曾接受氟嘧啶,奥沙利铂和伊立替康为基础的化疗,抗VEGF生物治疗,以及RAS野生型,抗EGFR治疗。•转移性胃或胃食管连接腺癌,至少用两种化疗方案进行治疗,包括氟嘧啶,铂,紫杉烷或伊立替康,以及HER2/neu靶向治疗。
剂量和给药
•推荐剂量:每天两次口服35mg/m2/剂,每天28天的第1天至第5天和第8天至第12天食用。
剂量形式和强度
片剂:•15mg三氟尿苷/6.14mg tipiracil•20mg三氟尿苷/8.19mg tipiracil禁忌症没有。
警告和注意事项
•严重的骨髓抑制:在每个周期的第15天之前和之后获得全血计数。根据建议扣留并恢复下一个较低的LONSURFdosage。•胚胎 - 胎儿毒性:可能导致胎儿伤害。告知女性对胎儿潜在风险的生殖潜力并使用有效的感染。
不良反应
最常见的不良反应或实验室异常(≥10%)区域性贫血,中性粒细胞减少,疲劳/虚弱,恶心,血小板减少,萎缩,腹泻,呕吐和发热。
用于特定人群
•哺乳期:建议不要母乳喂养。•老年人使用:3岁或4岁的中性粒细胞减少症和血小板减少症以及第3级贫血症在65岁或以上的患者中更常见。•肝功能损害:不要在基线中度或严重肝功能损害的患者中启动LONSURF。
包装提供/存储和处理
LONSURF 15mg/6.14mg片剂以白色,双凸面,圆形,薄膜包衣片剂形式提供,一面印有'15',另一面印有'102'和'15mg',灰色墨水。在以下介绍中,这些药片采用HDPE瓶包装,并带有儿童防护罩:•20计数:NDC 64842-1025-1•40计数:NDC 64842-1025-2•60计数:NDC 64842-1025-3LONSURF 20mg/8.19mg片剂以浅红色,双凸面,圆形薄膜包衣片剂形式提供,一面印有'20',另一面印有'102'和'20mg',灰色墨水。在以下介绍中,这些药片采用HDPE瓶包装,并带有儿童防护罩:•20计数:NDC 64842-1020-1•40计数:NDC 64842-1020-2•60计数:NDC 64842-1020-3储存在20°C至25°C(68°F至77°F);允许从15°C到30°C(59°F到50°C)的偏移86°F)[见USP受控室温]。LONSURF是一种细胞毒性药物。遵循适用的特殊处理和处理程序.1如果存放在原瓶外,30天后丢弃。
英文版说明书
SAFETY INFORMATIONWARNINGS AND PRECAUTIONSSevere Myelosuppression: LONSURF caused severe and life‑threatening myelosuppression (Grade 3‑4) consisting of neutropenia (38%), anemia (18%), thrombocytopenia (5%), and febrile neutropenia (3%). Two patients (0.2%) died due to neutropenic infection. A total of 12% of LONSURF‑treated patients received granulocyte‑colony stimulating factors. Obtain complete blood counts prior to and on day 15 of each cycle of LONSURF and more frequently as clinically indicated. Withhold LONSURF for febrile neutropenia, absolute neutrophil count less than 500/mm3, or platelets less than 50,000/mm3. Upon recovery, resume LONSURF at a reduced dose as clinically indicated.Embryo‑Fetal Toxicity: LONSURF can cause fetal harm when administered to a pregnant woman. Advise pregnant women of the potential risk to the fetus. Advise females of reproductive potential to use effective contraception during treatment and for at least 6 months after the final dose.USE IN SPECIFIC POPULATIONSLactation: It is not known whether LONSURF or its metabolites are present in human milk. There are no data to assess the effects of LONSURF or its metabolites on the breast‑fed infant or the effects on milk production. Because of the potential for serious adverse reactions in breast‑fed infants, advise women not to breastfeed during treatment with LONSURF and for 1 day following the final dose.Male Contraception: Because of the potential for genotoxicity, advise males with female partners of reproductive potential to use condoms during treatment with LONSURF and for at least 3 months after the final dose.Geriatric Use: Patients 65 years of age or over who received LONSURF had a higher incidence of the following compared to patients younger than 65 years: Grade 3 or 4 neutropenia (46% vs 32%), Grade 3 anemia (22% vs 16%), and Grade 3 or 4 thrombocytopenia (7% vs 4%).Hepatic Impairment: Do not initiate LONSURF in patients with baseline moderate or severe (total bilirubin greater than 1.5 times ULN and any AST) hepatic impairment. Patients with severe hepatic impairment (total bilirubin greater than 3 times ULN and any AST) were not studied. No adjustment to the starting dose of LONSURF is recommended for patients with mild hepatic impairment.Renal Impairment: No adjustment to the starting dosage of LONSURF is recommended in patients with mild or moderate renal impairment (CLcr of 30 to 89 mL/min). Patients with severe renal impairment (CLcr < 30 mL/min) were not studied.ADVERSE REACTIONSMost Common Adverse Drug Reactions in Patients Treated With LONSURF (≥5%): The most common adverse drug reactions in LONSURF‑treated patients vs placebo‑treated patients with mCRC, respectively, were asthenia/fatigue (52% vs 35%), nausea (48% vs 24%), decreased appetite (39% vs 29%), diarrhea (32% vs 12%), vomiting (28% vs 14%), infections (27% vs 16%), abdominal pain (21% vs 18%), pyrexia (19% vs 14%), stomatitis (8% vs 6%), dysgeusia (7% vs 2%), and alopecia (7% vs 1%). In metastatic gastric cancer or gastroesophageal junction (GEJ), the most common adverse drug reactions respectively were, nausea (37% vs 32%), decreased appetite (34% vs 31%), vomiting (25% vs 20%), infections (23% vs 16%) and diarrhea (23% vs 14%).Pulmonary emboli occurred more frequently in LONSURF‑treated patients compared to placebo: (2% vs 0%) in mCRC and (3% vs 2%) in metastatic gastric cancer and GEJ.Interstitial lung disease (0.2%), including fatalities, has been reported in clinical studies and clinical practice settings in Asia.Laboratory Test Abnormalities in Patients Treated With LONSURF: Laboratory test abnormalities in LONSURF‑treated patients vs placebo‑treated patients with mCRC, respectively, were anemia (77% vs 33%), neutropenia (67% vs 1%), and thrombocytopenia (42% vs 8%). In metastatic gastric cancer or GEJ, the test abnormalities, respectively, were neutropenia (66% vs 4%), anemia (63% vs 38%), and thrombocytopenia (34% vs 9%).
用药温馨提示:当您服用此药物时,需定期接受医疗专业人士的检查,以便随时针对其药效、副作用等情况进行监测。本网站所包含的信息旨在为患者提供帮助,不能代替医学建议和治疗。
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 曲氟尿苷盐酸/替吡嘧啶复方片Lonsurf Tablets 15/6.14mg说明书
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 曲氟尿苷盐酸/替吡嘧啶复方片Lonsurf Tablets 15/6.14mg说明书