阿雷替尼胶囊alectinib(Alecensa Capsules 150mg)
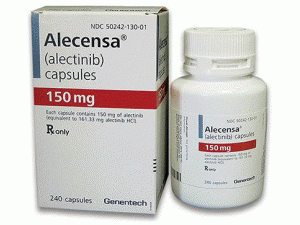 产地国家:美国
产地国家:美国
处方药:是
所属类别: 150毫克/胶囊 240胶囊/瓶
包装规格: 150毫克/胶囊 240胶囊/瓶
计价单位:瓶
生产厂家英文名:GENENTECH USA
原产地英文商品名:ALECENSA 150MG CAP DPSH 240/EA
原产地英文药品名:ALECTINIB
中文参考商品译名:Alecensa胶囊 150毫克/胶囊 240胶囊/瓶
中文参考药品译名:盐酸阿雷替尼
曾用名:AP26113,艾乐替尼,阿雷替尼
简介
近日,美国食品和药物管理局(FDA)已授予口服靶向抗癌药Alecensa(alectinib)第2个突破性药物资格(BTD),用于之前未接受ALK抑制剂治疗的晚期间变性淋巴瘤激酶(ALK)阳性非小细胞肺癌(NSCLC)成人患者。Alecensa是一种ALK抑制剂,之前,FDA于2013年首次授予该药突破性药物资格,用于既往接受辉瑞口服靶向抗癌药Xalkori(crizotinib,克唑替尼)治疗后病情进展的ALK阳性NSCLC患者。
批准日期:2016年10月8日
公司:GenentechALECENSA(阿雷替尼[alectinib])胶囊,用于口服
美国最初批准:2015年
最近的重大变化适应症和用法:11/2017剂量和用量:11/2017剂量和用法:06/2018
警告和注意事项:11/2017
作用机制:Alectinib是一种靶向ALK和RET的酪氨酸激酶抑制剂。 在非临床研究中,alectinib抑制ALK磷酸化和ALK介导的下游信号蛋白STAT3和AKT的激活,并降低了携带ALK融合,扩增或激活突变的多种细胞系中的肿瘤细胞活力。 alectinib的主要活性代谢物M4显示出相似的体外效力和活性。Alectinib和M4显示出针对ALK酶的多种突变形式的体外和体内活性,包括在克唑替尼上进展的患者中在NSCLC肿瘤中鉴定的一些突变。在植入携带ALK融合的肿瘤的小鼠模型中,施用alectinib导致抗肿瘤性和延长的存活,包括在用ALK驱动的肿瘤细胞系颅内植入的小鼠模型中。
适应症和用法:ALECENSA是一种激酶抑制剂,用于治疗患有促炎性淋巴瘤激酶(ALK)阳性转移性非小细胞肺癌(NSCLC)的患者,如FDA批准的试验所检测。
剂量和给药:每日口服600毫克600毫克。用食物管理ALECENSA。
剂量形式和强度胶囊:150mg
禁忌症:没有。
警告和注意事项:
肝毒性:在治疗的前3个月内每2周监测一次肝脏实验室检查,然后每月检查一次,根据临床指示,对发生转氨酶和胆红素升高的患者进行更频繁的检测。如果严重的ALT,AST或胆红素升高,请停止,然后减少剂量,或永久停止ALECENSA。
间质性肺病(ILD)/肺炎:在确诊患有ILD /肺炎的患者中立即扣留ALECENSA,如果没有其他可能的ILD/肺炎原因被识别,则会永久性地停止。
肾功能损害:扣留ALECENSA用于严重肾功能损害,然后在恢复或永久停用时以减少的剂量恢复ALECENSA。
心动过缓:定期监测心率和血压。如果有症状,扣留ALECENSA然后减少剂量,或永久停止。
严重肌痛和肌酸磷酸激酶(CPK)升高:在治疗的第一个月和报告肌肉疼痛,压痛或虚弱的患者中评估CPK每2周。如果出现严重的CPKelevations,请扣留,然后恢复或减少剂量。
胚胎 - 胎儿毒性:ALECENSA可导致胎儿伤害。告知女性对胎儿潜在风险的生殖潜力并使用有效的感染。
不良反应:最常见的不良反应(发生率≥20%)是疲劳,便秘,水肿,肌痛和贫血。
用于特定人群:哺乳期,不要母乳喂养。
包装提供/存储和处理:硬胶囊,白色150毫克胶囊,“ALE”印在黑色墨水盖上,“150毫克”印有黑色墨水,可用于:每瓶240粒:储存和稳定性:不要储存在30°C(86°F)以上。 存放在原装容器中,以防止光线和水分。
英文版说明书
download/pdf/alecensa_prescribing.pdfFDA Approves Alecensa (Alectinib) for Non-Small Cell Lung CancerThe US Food and Drug Administration (FDA) has approved Alecensa (alectinib) to treat people with advanced ALK-positive non-small cell lung cancer (NSCLC) that has become worse after treatment with another targeted therapy drug called Xalkori (crizotinib). It’s also for people who could not tolerate taking Xalkori in the first place.About 5% of NSCLCs have been found to have a rearrangement in a gene called ALK. This change is most often seen in non-smokers (or light smokers) who have the adenocarcinoma subtype of NSCLC. Doctors may test cancers for changes in the ALK gene to see if drugs that target this change may help them.Alecensa is taken by mouth. It is thought to work by blocking the activity of the ALK protein, which may prevent NSCLC cells from growing and spreading. In metastatic cancer, the disease spreads to new parts of the body. In ALK-positive NSCLC patients, the brain is a common place for the disease to spread.“Today’s approval provides a new therapy for a group of patients who would have few treatment options once their disease no longer responds to treatment with Xalkori,” said Richard Pazdur, MD, director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug eva luation and Research, in a statement. “In addition to the primary effect on tumors in the lung, Alecensa clinical trials provide evidence of an effect on tumors that had spread to the brain, which is an important effect for clinicians to understand.”The FDA based its approval on 2 clinical trials of people with metastatic ALK-positive NSCLC whose disease could not be controlled with Xalkori. Study participants received Alecensa twice a day. In the first study, 38% of participants saw a partial shrinkage of their lung tumors, an effect that lasted for an average of 7.5 months. In the second study, 44% of participants saw a partial shrinkage of their lung tumors, lasting for an average of 11.2 months. The trials also looked at Alecensa’s effect on tumors that had spread to the brain. Of the participants who had measureable brain metastases, 61% saw a complete or partial reduction in their brain tumors, lasting an average of 9.1 months.The most common side effects of Alecensa are fatigue, constipation, swelling (edema) and muscle pain (myalgia). Alecensa may cause serious side effects, including liver problems, severe or life-threatening inflammation of the lungs, very slow heartbeats and severe muscle problems. Alecensa may make people burn more easily when out in the sun.Alecensa was approved using several FDA approaches designed to speed up the availability of drugs to treat serious diseases – breakthrough therapy designation, priority review, and accelerated approval. It was also granted orphan drug designation, which provides financial incentives to encourage the development of drugs for rare diseases.
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 阿雷替尼胶囊alectinib(Alecensa Capsules 150mg)