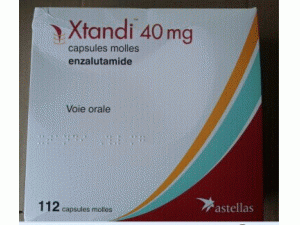
恩杂鲁胺胶囊enzalutamide(Xtandi 40mg capsules)
 产地国家:法国
产地国家:法国
处方药:是
所属类别: 40毫克/胶囊 112胶囊/盒
包装规格: 40毫克/胶囊 112胶囊/盒
计价单位:盒
生产厂家英文名:ASTELLAS
原产地英文商品名:Xtandi 40mg/cap 112caps/box
原产地英文药品名:ESCITALOPRAM
中文参考商品译名:Xtandi 40毫克/胶囊 112胶囊/盒
中文参考药品译名:恩杂鲁胺
简介
部份中文恩杂鲁胺处方资料(仅供参考)
英文名:enzalutamide
商标名:XTANDI
中文名:恩杂鲁胺
生产商:安斯泰来
药品简介:XTANDI(恩杂鲁胺[enzalutamide])胶囊为,口服使用批准日期:2012。
作用机制:Enzalutamide是一种雄激素受体抑制剂作用在雄激素受体信号通路不同步骤。曾证明Enzalutamide与雄激素竞争性抑制结合至雄激素受体和抑制雄激素受体核易位和与DNA相互作用。一个主要代谢物,N-去甲基enzalutamide,表现出与enzalutamide相似体外活性。在体外Enzalutamide减低增殖和诱发前列腺癌细胞的细胞死亡,和在小鼠前列腺癌异种移植模型中减小肿瘤体积。
适应证和用途:XTANDI是一种雄激素受体抑制剂适用于治疗有转移去势耐受前列腺癌既往曾接受多西他奇[docetaxel]患者。
剂量和给药方法:XTANDI 160 mg(四40 mg胶囊)口服给药每天1次。吞服整个胶囊。XTANDI可与或无食物服用。
剂型和规格:胶囊,40mg
禁忌证:妊娠
警告和注意事项:接受XTANDI患者0.9%发生癫痫发作。没有曾有癫痫发作患者,在有癫痫发作诱发因素患者,或同时使用药物可能降低癫痫发作阈患者中使用XTANDI的临床试验经验。
不良反应:最常见不良反应(≥5%)是虚弱/疲劳,背痛,腹泻,关节痛,潮热,外周血水肿,肌肉骨骼痛,头痛,上呼吸道感染,肌肉无力,眩晕,失眠,下呼吸道感染,脊髓压迫症和马尾神经综合征,血尿,感觉异常,焦虑,和高血压。
药物相互作用:(1)避免强CYP2C8抑制剂,因为它们可能增加对XTANDI血浆暴露。如需要共同给药,减低XTANDI剂量。(2)避免强或中度CYP3A4或CYP2C8 诱导剂因它们可能改变对XTANDI血浆暴露。(3)避免CYP3A4,CYP2C9和CYP2C19底物有狭窄治疗指数,因XTANDI可能减低这些药物的血浆暴露。如XTANDI是与华法林(CYP2C9底物)共同给药,进行附加的INR监测。
英文版说明书
XTANDI (enzalutamide) capsules is indicated for the treatment of patients with metastatic castration-resistant prostate cancer (CRPC).Important Safety InformationContraindicationsXTANDI is not indicated for women. XTANDI can cause fetal harm and potential loss of pregnancy.Warnings and PrecautionsSeizure occurred in 0.5% of patients receiving XTANDI in clinical studies. In a study of patients with predisposing factors, seizures were reported in 2.2% of patients. See section 5.1 of the Prescribing Information for the list of predisposing factors. It is unknown whether anti-epileptic medications will prevent seizures with XTANDI. Permanently discontinue XTANDI in patients who develop a seizure during treatment.Posterior Reversible Encephalopathy Syndrome (PRES) In post approval use, there have been reports of PRES in patients receiving XTANDI. PRES is a neurological disorder which can present with rapidly evolving symptoms including seizure, headache, lethargy, confusion, blindness, and other visual and neurological disturbances, with or without associated hypertension. A diagnosis of PRES requires confirmation by brain imaging, preferably MRI. Discontinue XTANDI in patients who develop PRES.Adverse ReactionsThe most common adverse reactions (≥ 10%) that occurred more commonly (≥ 2% over placebo) in the XTANDI patients from the two placebo-controlled clinical trials were asthenia/fatigue, back pain, decreased appetite, constipation, arthralgia, diarrhea, hot flush, upper respiratory tract infection, peripheral edema, dyspnea, musculoskeletal pain, weight decreased, headache, hypertension, and dizziness/vertigo. In the bicalutamide-controlled study of chemotherapy-naïve patients, the most common adverse reactions (≥ 10%) reported in XTANDI patients were asthenia/fatigue, back pain, musculoskeletal pain, hot flush, hypertension, nausea, constipation, upper respiratory tract infection, diarrhea, and weight loss.In the placebo-controlled study of patients taking XTANDI who previously received docetaxel, Grade 3 and higher adverse reactions were reported among 47% of XTANDI patients and 53% of placebo patients. Discontinuations due to adverse events were reported for 16% of XTANDI patients and 18% of placebo patients. In the placebo-controlled study of chemotherapy-naïve patients, Grade 3-4 adverse reactions were reported in 44% of XTANDI patients and 37% of placebo patients. Discontinuations due to adverse events were reported for 6% of both study groups. In the bicalutamide-controlled study of chemotherapy-naïve patients, Grade 3-4 adverse reactions were reported in 38.8% of XTANDI patients and 37.6% of bicalutamide patients. Discontinuations due to adverse events were reported for 7.6% of XTANDI patients and 6.3% of bicalutamide patients.Lab Abnormalities: In the two placebo-controlled trials, Grade 1-4 neutropenia occurred in 15% of XTANDI patients (1% Grade 3-4) and 6% of placebo patients (0.5% Grade 3-4). Grade 1-4 thrombocytopenia occurred in 6% of XTANDI patients (0.3% Grade 3-4) and 5% of placebo patients (0.5% Grade 3-4). Grade 1-4 elevations in ALT occurred in 10% of XTANDI patients (0.2% Grade 3-4) and 16% of placebo patients (0.2% Grade 3-4). Grade 1-4 elevations in bilirubin occurred in 3% of XTANDI patients (0.1% Grade 3-4) and 2% of placebo patients (no Grade 3-4).Infections: In the study of patients taking XTANDI who previously received docetaxel, 1% of XTANDI patients compared to 0.3% of placebo patients died from infections or sepsis. In the study of chemotherapy-naïve patients, 1 patient in each treatment group (0.1%) had an infection resulting in death.Falls (including fall-related injuries) occurred in 9% of XTANDI patients and 4% of placebo patients in the two placebo-controlled trials. Falls were not associated with loss of consciousness or seizure. Fall-related injuries were more severe in XTANDI patients, and included non-pathologic fractures, joint injuries, and hematomas.Hypertension occurred in 11% of XTANDI patients and 4% of placebo patients in the two placebo-controlled trials. No patients experienced hypertensive crisis. Medical history of hypertension was balanced between arms. Hypertension led to study discontinuation in < 1% of patients in each arm.Drug InteractionsEffect of Other Drugs on XTANDI Avoid strong CYP2C8 inhibitors, as they can increase the plasma exposure to XTANDI. If co-administration is necessary, reduce the dose of XTANDI.Avoid strong CYP3A4 inducers as they can decrease the plasma exposure to XTANDI. If co-administration is necessary, increase the dose of XTANDI.Effect of XTANDI on Other Drugs Avoid CYP3A4, CYP2C9, and CYP2C19 substrates with a narrow therapeutic index, as XTANDI may decrease the plasma exposures of these drugs. If XTANDI is co-administered with warfarin (CYP2C9 substrate), conduct additional INR monitoring.
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 恩杂鲁胺胶囊enzalutamide(Xtandi 40mg capsules)