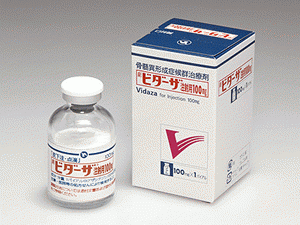
阿扎胞苷冻干粉注射剂Azacitidine(Vidaza 100mg)
 产地国家:日本
处方药:是
所属类别: 100毫克/瓶
包装规格: 100毫克/瓶
计价单位:瓶
生产厂家英文名:nippon-shinyaku
原产地英文商品名:VIDAZA(ビダーザ) 100mg/Vial
原产地英文药品名:Azacitidine
中文参考商品译名:维达扎(ビダーザ) 100毫克/瓶
中文参考药品译名:阿扎胞苷
曾用名:阿扎胞苷、5-氮杂胞苷、5-氮杂胞嘧啶核苷、氮胞苷、氮杂胞苷、Ladakamycin
产地国家:日本
处方药:是
所属类别: 100毫克/瓶
包装规格: 100毫克/瓶
计价单位:瓶
生产厂家英文名:nippon-shinyaku
原产地英文商品名:VIDAZA(ビダーザ) 100mg/Vial
原产地英文药品名:Azacitidine
中文参考商品译名:维达扎(ビダーザ) 100毫克/瓶
中文参考药品译名:阿扎胞苷
曾用名:阿扎胞苷、5-氮杂胞苷、5-氮杂胞嘧啶核苷、氮胞苷、氮杂胞苷、Ladakamycin
简介:
部份中文阿扎胞苷处方资料(仅供参考)英文名:Azacitidine商品名:Vidaza for Injection中文名:阿扎胞苷冻干粉注射剂生产商:日本新药公司ビダーザ注射用100mg药用类别名称骨髓增生异常综合征治疗剂批准日期:2011年3月商標名Vidaza for Injection 100mg一般名アザシチジン(Azacitidine)(JAN)化学名4-Amino-1-β-D-ribofuranosyl-1,3,5-triazin-2(1H)-one分子式C8H12N4O5分子量244.20化学構造式性状白色到细灰色固体。本品易溶于二甲基亚硫酸, 不易溶于水或 n-甲基吡咯烷酮, 几乎不溶于乙醇 (99.5)。熔点约 227°c (拆卸)批准条件由于日本的临床试验病例极为有限, 在制造和销售之后, 直到积累了一定数量的病例数据, 通过对所有病例进行使用等级调查, 掌握使用这种药物的患者的背景信息, 在早期阶段收集有关该制剂安全性和有效性的数据, 并采取必要措施正确使用该制剂。 作用机制:这种药物通过被纳入DNA和RNA, 主要抑制蛋白质合成, 显示细胞的杀灭作用。 此外, 在MDS中, 报告了抑制癌症基因启动子区域dna的高甲基化和抑制癌症抑制基因的情况, 通过抑制DNA 的甲基化, 将 Azacytidine纳入DNA, 另据报道, 有可能显示细胞增殖的抑制作用。 药理作用:(1) Azacytidine 在体外试验中显示从 MDS 迁移到急性髓系白血病的患者SKM-1细胞系生长抑制作用。(2) 阿司替丁, 对nodw scid小鼠进行皮下注射的SKM-1细胞系, 显示出肿瘤生长的抑制作用。 适应症:骨髓增生异常综合症。 用法与用量:通常情况下, 成人 75 Mg/mm2 作为 azacytidine (体表面积), 每天一次, 为期7天的皮下注射或10分钟的静脉输液, 至3周休息。这是一个周期, 也是反复的管理。另外, 根据患者的病情酌情用药。 包装:注射100毫克: 1瓶 完整说明资料附件:https://www.info.pmda.go.jp/go/pack/4291419D1026_1_05/英文版说明书:
AZACITIDINE CELGENE CORPORATION 59572-0102-01Vidaza (azacitidine)VIDAZA is indicated for treatment of patients with the following French-American-British (FAB) myelodysplastic syndrome subtypes: refractory anemia (RA) or refractory anemia with ringed sideroblasts (RARS) (if accompanied by neutropenia or thrombocytopenia or requiring transfusions), refractory anemia with excess blasts (RAEB), refractory anemia with excess blasts in transformation (RAEB-T), and chronic myelomonocytic leukemia (CMMoL).IMPORTANT SAFETY INFORMATION•VIDAZA is contraindicated in patients with a known hypersensitivity to azacitidine or mannitol and in patients with advanced malignant hepatic tumors.•In Studies 1 and 2, the most commonly occurring adverse reactions by SC route were nausea (70.5%), anemia (69.5%), thrombocytopenia (65.5%), vomiting (54.1%), pyrexia (51.8%), leukopenia (48.2%), diarrhea (36.4%), injection site erythema (35.0%), constipation (33.6%), neutropenia (32.3%), and ecchymosis (30.5%). Other adverse reactions included dizziness (18.6%), chest pain (16.4%), febrile neutropenia (16.4%), myalgia (15.9%), injection site reaction (13.6%), and malaise (10.9%). In Study 3, the most common adverse reactions by IV route also included petechiae (45.8%), weakness (35.4%), rigors (35.4%), and hypokalemia (31.3%).•In Study 4, the most commonly occurring adverse reactions were thrombocytopenia (69.7%), neutropenia (65.7%), anemia (51.4%), constipation (50.3%), nausea (48.0%), injection site erythema (42.9%), and pyrexia (30.3%). The most commonly occurring Grade 3/4 adverse reactions were neutropenia (61.1%), thrombocytopenia (58.3%), leukopenia (14.9%), anemia (13.7%) and febrile neutropenia (12.6%).•Because treatment with VIDAZA is associated with anemia, neutropenia and thrombocytopenia, complete blood counts should be performed as needed to monitor response and toxicity, but at a minimum, prior to each dosing cycle.•Because azacitidine is potentially hepatotoxic in patients with severe preexisting hepatic impairment, caution is needed in patients with liver disease. In addition, azacitidine and its metabolites are substantially excreted by the kidneys and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, it may be useful to monitor renal function.•VIDAZA may cause fetal harm when administered to a pregnant woman. Women of childbearing potential should be apprised of the potential hazard to the fetus. Men should be advised not to father a child while receiving VIDAZA.•Nursing mothers should be advised to discontinue nursing or the drug, taking into consideration the importance of the drug to the mother.Please see full Prescribing Information.用药温馨提示:当您服用此药物时,需定期接受医疗专业人士的检查,以便随时针对其药效、副作用等情况进行监测。本网站所包含的信息旨在为患者提供帮助,不能代替医学建议和治疗。
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 阿扎胞苷冻干粉注射剂Azacitidine(Vidaza 100mg)
药品价格查询,专业药品查询网站,药品说明书查询,药品比价 » 阿扎胞苷冻干粉注射剂Azacitidine(Vidaza 100mg)